 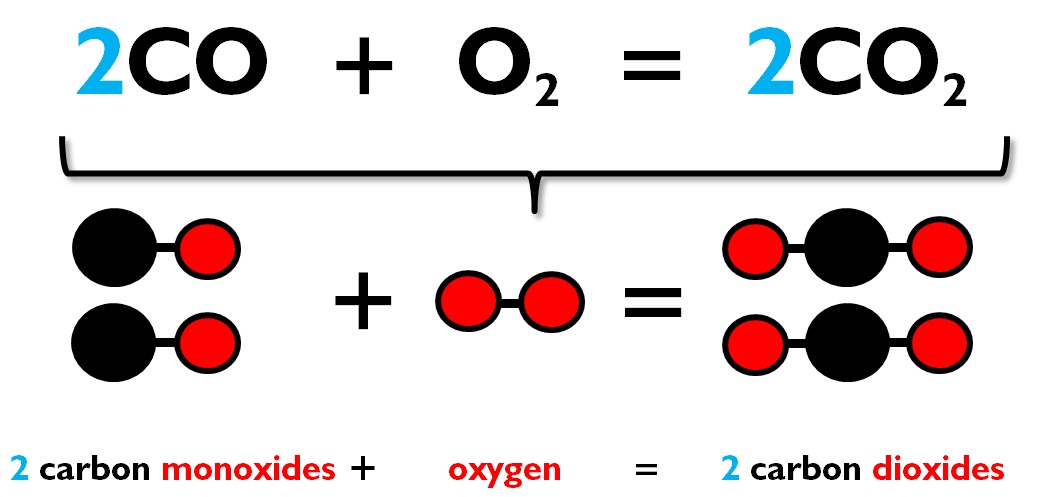
It belongs to group 14 of the periodic table. It is nonmetallic and tetravalent-its atom making four electrons available to form covalent chemical bonds. The trends in chemical reactivity of Carbon can be understood even by assessing the formation of present day-to-day compounds of Carbon like carbon dioxide and other compounds.−4, −3, −2, −1, 0, +1, +2, +3, +4 (a mildly acidic oxide)Ĭarbon (from Latin carbo 'coal') is a chemical element with the symbol C and atomic number 6. The trends in chemical reactivity are quite apparent when pointed out and also a bit revealing of the true nature of the element. 
Carbon should be appreciated as an element more, and that cannot be made any more apparent other than what has been discussed here. This article will be beneficial for understanding the trends in detail and also in a simple language. ConclusionĪll in all, the trends in the chemical reactivity of Carbon have been quite thoroughly and efficiently explained. Although it is known to react with a whole array of elements, Carbon is very weak in terms of reactivity in normal conditions. The ability of carbon and carbon bonds to be strong and stable is a big advantage of Carbon as it can form such chains in infinite numbers of carbon bonds to form compounds, and it is due to this nature of Carbon that organic chemistry has such an ample research base. But Carbon is a fairly viable element to react with because of its tetravalency and also its self interlinking nature. We already know that Carbon has difficulty in reacting with other substances due to the extreme conditions required for the reactions to take place, like the formation of carbon oxides and metal carbides. 
As further right and to the top one moves in case of non-metals, more the reactivity because of the increasing values of electronegativity, which facilitates the smooth exchange of electrons. In the perspective of groups, the reactivity decreases as one goes down a group. Reactivity increases as one moves from left to right in the case of non-metals with respect to periods. In the case of non-metals, it is almost the vice versa that of metals. The further down and further left one goes, the easier it becomes for elements to react by the exchange of electrons because the reactivity becomes very high. By group, reactivity increases as one goes down a group. In the case of metals, by period, reactivity decreases as one moves from left to right. Usual Trends in Chemical Reactivity in the Periodic Table As for chemical reactivity, there are variations in trends for metals and non-metals. They can also be termed periodic trends in chemical reactivity. Trends in chemical reactivity refer to the recurrence of features of properties, both physical and chemical, in the chemical perspective of elements on the periodic table, based on their ability to react with other substances and how they behave after the reaction. If observed acutely, one can find some regularities in the chemical reactivity sector of Carbon, which are also called trends in chemical reactivity. Chemically speaking, Carbon does not readily or spontaneously react with other substances because of its stability and also because of the extreme conditions required for it to react with other substances. In terms of boiling point and melting point, Carbon has a whopping melting point of 3652☌ and an extreme boiling point of 4827☌.Ĭarbon-based compounds form the foundation of all living life forms on the surface of the earth as we know it. Represented by the letter C, Carbon has an atomic number of 6 and has an electronic configuration of 1s2 2s2 2p2. Before going into the chemical reactivity of Carbon and its trends, let us have a look at some of the properties of Carbon. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
