 Note that the O2 result on the left is the NET of the (3/2)O2 on the left, and the (1/2) O2 on the right, as addends. Get the detailed answer: the vaules of delta h and delta s rxn for the reaction 2 NO(g) O2(g) -> 2 NO2(g) are -147 j/k and -12.0 kj. When delta H and delta S are both positive, then delta G would be positive at high temperatures and delta G would be negative at low temperatures. delta.H = -395.2kJ (note that we don't say "per mole" which would be ambigous, rather just leave it as is and know that it means for the equation as written)Īdding: S O2 SO2. When the heat is supplied, the enthalpy of the reaction is positive and hence, the reaction is endothermic but when the heat is released, the enthalpy of the reaction is negative and hence, the. Now you can write your equations and add them: Why: the sign flips when you flip the equation, and the enthalpy number drops by 0.5 when you scale the reaction by 0.5. 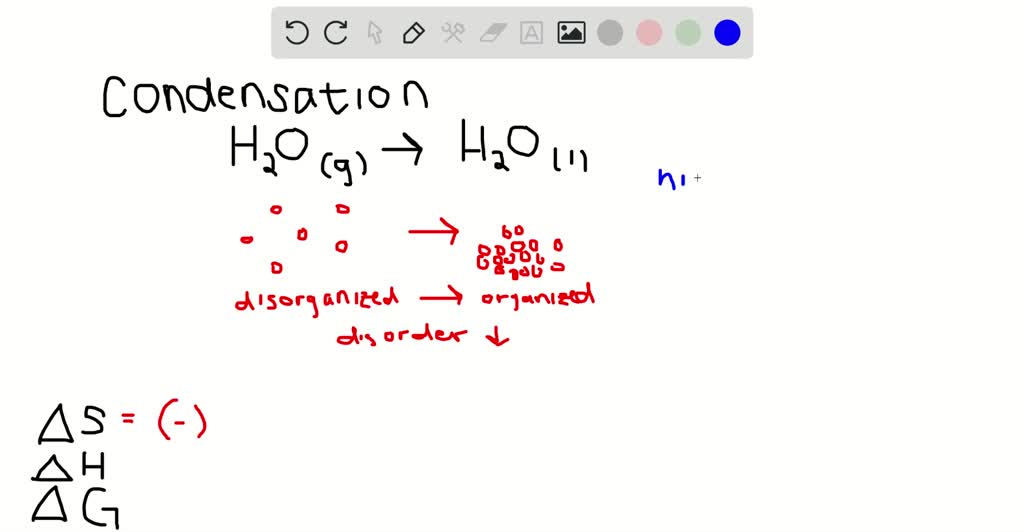
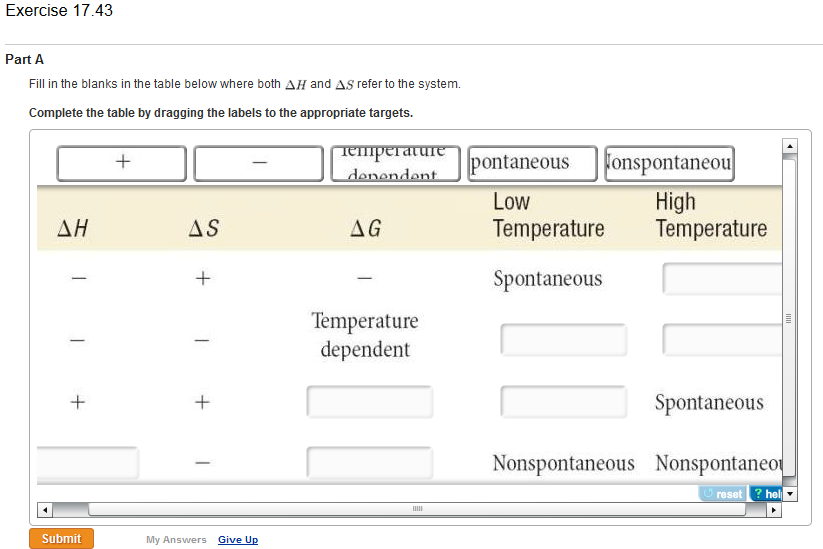
Turn around your second input equation, and multiply it throughout by the factor (1/2): Calculate Delta Hrxn for the following reaction: CaO(s) CO2(g)->CaCO3(s) Use the following reactions and given delta H values: Ca(s) CO2(g) 12O2(g)->CaCO3(s), delta H -812.8 kJ 2Ca(s) O2(g)->2 Calculate Delta Gcirc for the following reaction at 25circ C. But you have that SO3 on the right! So, prepare to get rid of it by making it appear on the left in an equation, so as to cancel the one on the right when you add the 2 equations. Now, are you lacking any reactants on the left? Not really. So pick an equation from your input list with an S on the left: I am trying to solve a few questions and one of those is : For the process, H2O (g) 2 H (g) O (g), one would expect H to be negative and S to be positive. Look at your desired equation: S O2 = SO2 For elements it's always zero, even the o2 or the diatomics. Calculate Delta Hrxn for the following reaction: CaO(s) CO2(g)->CaCO3(s) Use the following reactions and given delta H values: Ca(s) CO2(g) 12O2(g)->CaCO3(s), delta H -812.8 kJ 2Ca(s) O2(g)->2 Calculate Delta S for the reaction 2H2(g) O2(g) arrow 2H2O(g) where Delta S for H2(g), O2(g), and H2O(g), respectively, is 130.6, 205. It actually is a process going from its elements reforming that. For compounds, when we're talking of the delta H, it's a formation of that. Delta H is looking at transfer of enthalpy/heat-Delta S is looking at randomness/disorder CASE 1-The universe prefers exothermic reactions, because it lowers the energy of the system and if you lower the energy. So if we have an element, elements have a delta H of zero, always. There are very many routes to accomplish the same thing here's one of them. We know if Delta G >0 reaction is non spontaneous We know if Delta G <0 reaction is spontaneous We know Delta G 0 at equilibrium But now lets define that tin terms of Delta H and S. 
Thus although the free energy always falls when a gas expands or a chemical reaction takes place spontaneously, there need be no compensating increase in energy anywhere else.You want to add your equations so that you get your intended final equation, right?
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
